China’s inactivated COVID-19 vaccine entered the second phase of clinical trial on Friday, according to its developers.
The Chinese firm said it is one of the first organisations to have led the development of a vaccine for the novel coronavirus. A state-owned enterprise, Sinopharm produces more than 80% of China’s mandatory immunisation needs and played a leading role in fighting the Covid-19 crisis in China. Read now China Approves Third COVID-19 Vaccine For Clinical Trials, Invites Pakistan For Vaccine Test
The inactivated vaccine developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm) and the Wuhan Institute of Virology under the Chinese Academy of Sciences started its clinical trial on April 12.
Sinopharm said in an announcement that 96 persons in three age groups have received the vaccine in the first phase of clinical trial as of April 23. The vaccine has shown good safety so far and vaccine receivers are still under observation.
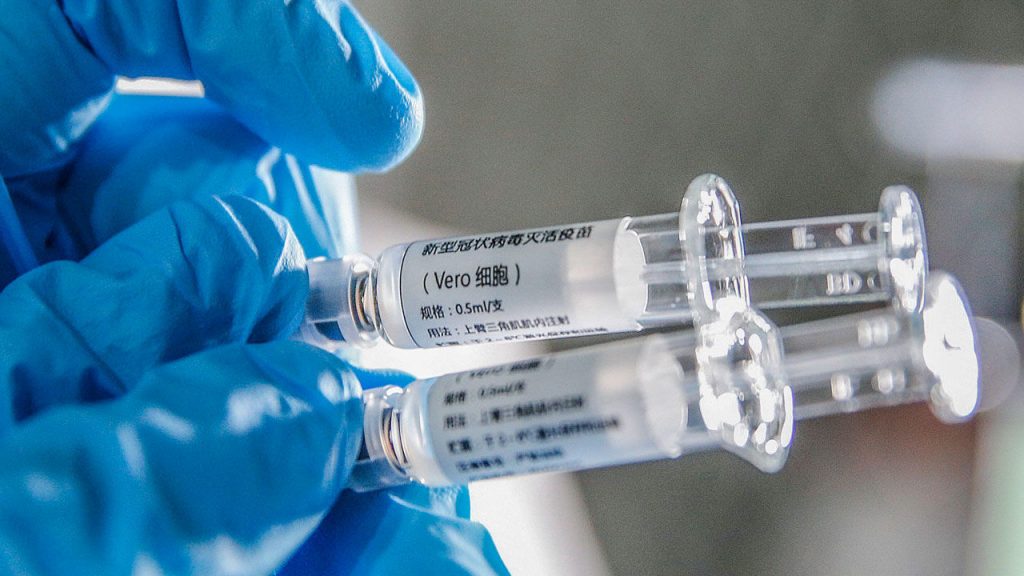
The randomized, double-blind and placebo-controlled clinical trials of the inactivated vaccine are conducted in Jiaozuo, central China’s Henan Province, and the second phase of clinical trial will focus on the vaccination procedure, according to Sinopharm.
Both the first and the second phase of clinical trials of the vaccine has been approved by the National Medical Products Administration through a green channel.
Sinopharm said the vaccine will also go through the third phase of the clinical trial, and it may take about one year to complete the clinical trial and finally reach the conclusion on the vaccine’s safety and efficacy.
China has approved three COVID-19 vaccine candidates for clinical trials. An adenovirus vector vaccine was the first COVID-19 vaccine in the world that has entered the second phase of clinical trial.
Read Also: China-Italy Connection Of CoronaVirus
Chinese pharmaceutical company has invited a Pakistani health institute to collaborate in its recently developed “inactivated” Covid-19 vaccine for clinical trials in Pakistan.
A letter received on Wednesday by Dr Amir Ikram, the head of the Islamabad-based National Institute of Health (NIH), from Li Can, the general manager of China Sinopharm International Corporation, stated, “Sinopharm considers that NIH has the necessary technical expertise and elements for conducting the clinical trial on recruited participants, through a nominated medical institution, under phase I and II protocol requirements and as per clinical trial guidelines.”

Also on Friday, 29 asymptomatic cases were reported. So far, 983 suspected asymptomatic cases, including 150 from abroad, were still under medical observation, it said.
The asymptomatic cases were a cause of concern as the government has lifted over two-month lockdown in Hubei and Wuhan after cases abated.
Asymptomatic cases refer to people who are tested positive for the coronavirus but develop no symptoms such as fever, cough or sore throat. They are infectious and pose a risk of spreading to others.
For latest updates, stay tuned to our Facebook page: Woman’s Era

